Harold Urey, born on this date in 1893, discovered “heavy hydrogen” and “heavy water”!
Imagine a glass of water saying, “Does this hydrogen make me look heavy?”
Of course, the answer is that certain hydrogen atoms and kinds of water don't look heavy, but they are in fact heavier than “normal.” In other words, they weigh more.
If you want to know why, you need to learn a bit about isotopes.
Each kind of element—like carbon or gold or iron or oxygen—has recognizable characteristics. It's the kind of element that easily combines with other elements—or it isn't very reactive. It's a metal, or it isn't. It's radioactive, or not; brittle, or not. At room temperature, a particular element may be a solid, liquid, or gas. It has a certain color.
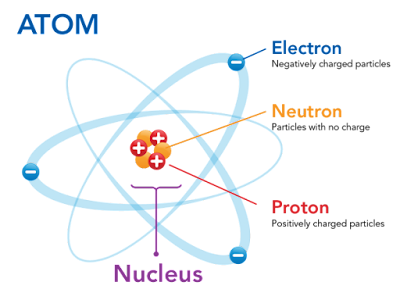
The thing that makes an element that element—in other words, what makes gold gold and oxygen oxygen—is the number of protons it has.
Gold always has 79 protons and generally has 79 electrons. Oxygen just has 8 protons (and, generally, 8 electrons). It's really hard to change the number of protons an atom has—that's what happens in the center of the sun and in nuclear reactors—but, if you do change the number of protons, you change the element. For example, take away one of gold's protons, you get platinum. Add a proton, you get mercury.
 |
The Periodic Table of the Elements is
organized in part by atomic number, which
is the number of protons each element has.
|
There aren't just protons and electrons in an atom; there are also neutrons. These particles are about a large as a proton but have no charge. Neutrons are necessary to help protons hang together despite their like charges.
The thing is, since neutrons have no charge, you can add them to an atom and not change the element. Most gold atoms have 117 or 118 neutrons—but if you add a few more or take a few away, you still have gold. You just have a different isotope of gold. It has the same characteristics (solid, metal, yellow colored), but it is heavier.
Forget about gold...I thought we were talking about hydrogen!
Hydrogen, which is the most common element, by far, in the universe, has a super simple structure: just one proton and one electron.
But some hydrogen atoms have one neutron as well. These hydrogen atoms act just like other hydrogen atoms (super light, super reactive, gaseous), but they are almost twice as heavy as normal hydrogen atoms.
 |
Different "flavors" of hydrogen.
The one labeled "protium" is by
far the most common! |
These heavier-than-normal atoms are called deuterium, or heavy hydrogen. About one out of every 6,400 hydrogen atoms in Earth's oceans is deuterium.
Discovery!
Back in the day, scientists didn't know why certain samples of materials were heavier than others, they didn't know the reason for the difference in weight; some theorized that it was caused by different numbers of “nuclear electrons.” (Remember, the difference is actually caused by more neutrons.) Scientists believed that hydrogen couldn't have isotopes. But an American chemist named Harold Urey was able to repeatedly distill liquid hydrogen and then—in 1931—to detect “heavy hydrogen” with a spectroscope.
And so it was that Urey and his coworkers were able to discover deuterium a year before scientists discovered neutrons—the correct explanation for deuterium!—and for his work, Urey won a Nobel Prize.
And heavy water tastes sweet! WARNING: Heavy water can be toxic. Only taste a tiny bit - and only with adult approval and supervision.










































.png)










